Original Paper
2022
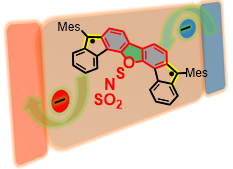
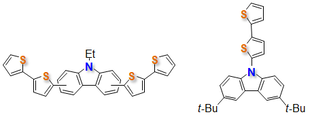
Medium Diradical Character, Small Hole and Electron Reorganization Energies and Ambipolar Transistors in Difluorenoheteroles
S. Mori, S. Moles Quintero, N. Tabata, R. Kishi, R. Gonzñález Núñez, A. Harbuzaru, R. Ponce Ortiz, J. Marín Beloqui, S. Suzuki, C. Kitamura, C. J. Gómez-García, Y. Dai, F. Negri, M. Nakano, S.-i. Kato, J. Casado
Angew. Chem. Int. Ed. 2022, 61, e202206680. DOI: 10.1002/anie.202206680
S. Mori, S. Moles Quintero, N. Tabata, R. Kishi, R. Gonzñález Núñez, A. Harbuzaru, R. Ponce Ortiz, J. Marín Beloqui, S. Suzuki, C. Kitamura, C. J. Gómez-García, Y. Dai, F. Negri, M. Nakano, S.-i. Kato, J. Casado
Angew. Chem. Int. Ed. 2022, 61, e202206680. DOI: 10.1002/anie.202206680
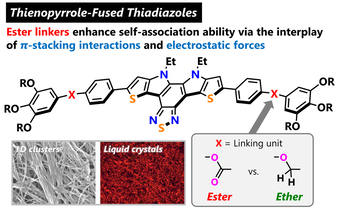
Augmented Self-Association by Electrostatic Forces in Thienopyrrole-Fused Thiadiazoles that Contain an Ester instead of an Ether Linker
Y. Naito, R. Moriguchi, C. Kitamura, T. Matsumoto, T. Yoshihara, T. Ishi-i, Y. Nagata, H. Takeshita, K. Yoshizawa, Y. Shiota, K. Suzuki, S.-i. Kato
Chem. Asian J. 2022, 17, e202101341. DOI: 10.1002/asia.202101341
Y. Naito, R. Moriguchi, C. Kitamura, T. Matsumoto, T. Yoshihara, T. Ishi-i, Y. Nagata, H. Takeshita, K. Yoshizawa, Y. Shiota, K. Suzuki, S.-i. Kato
Chem. Asian J. 2022, 17, e202101341. DOI: 10.1002/asia.202101341
2021
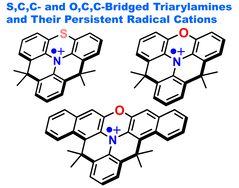
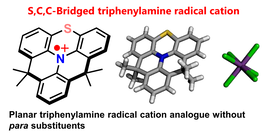
S,C,C- and O,C,C-Bridged Triarylamines and Their Persistent Radical Cations
S. Kataoka, S. Suzuki, Y. Shiota, K. Yoshizawa, T. Matsumoto, M. S. Asano, T. Yoshihara, C. Kitamura, S.-i. Kato
J. Org. Chem. 2021, 86, 12559. DOI: 10.1021/acs.joc.1c00969
S. Kataoka, S. Suzuki, Y. Shiota, K. Yoshizawa, T. Matsumoto, M. S. Asano, T. Yoshihara, C. Kitamura, S.-i. Kato
J. Org. Chem. 2021, 86, 12559. DOI: 10.1021/acs.joc.1c00969
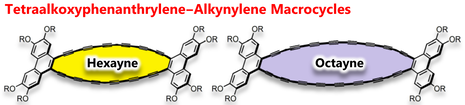
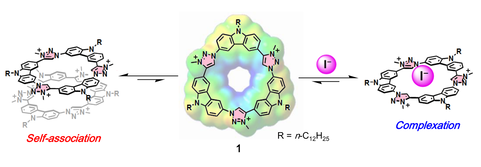
Arylene–Hexaynylene and –Octaynylene Macrocycles: Extending the Polyyne Chains Drives Self-Association by Enhanced Dispersion Force
S.-i. Kato, R. Kumagai, T. Abe, C. Higuchi, Y. Shiota, K. Yoshizawa, N. Takahashi, K. Yamamoto, Md Z. Hossain, K. Hayashi, T. Hirose, Y. Nakamura
Chem. Commun. 2021, 57, 555. DOI: 10.1039/D0CC07540J Highlighted as an outside front cover picture!
S.-i. Kato, R. Kumagai, T. Abe, C. Higuchi, Y. Shiota, K. Yoshizawa, N. Takahashi, K. Yamamoto, Md Z. Hossain, K. Hayashi, T. Hirose, Y. Nakamura
Chem. Commun. 2021, 57, 555. DOI: 10.1039/D0CC07540J Highlighted as an outside front cover picture!
2020
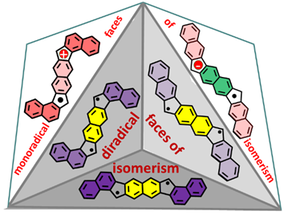
Monoradicals and Diradicals of Dibenzofluoreno[3,2-b]fluorene Isomers: Mechanisms of Electronic Delocalization
H. Hayashi, J. E. Barker, A. C. Valdivia, R. Kishi, S. N. MacMillan, C. J. Gómez-García, H. Miyauchi, Y. Nakamura, M. Nakano, S.-i. Kato, M. M. Haley, J. Casado.
J. Am. Chem. Soc. 2020, 142, 20444. DOI: 10.1021/jacs.0c09588
H. Hayashi, J. E. Barker, A. C. Valdivia, R. Kishi, S. N. MacMillan, C. J. Gómez-García, H. Miyauchi, Y. Nakamura, M. Nakano, S.-i. Kato, M. M. Haley, J. Casado.
J. Am. Chem. Soc. 2020, 142, 20444. DOI: 10.1021/jacs.0c09588
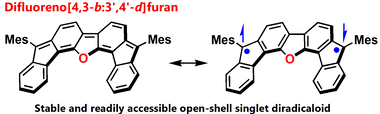
Open-Shell Singlet Diradicaloid Difluoreno[4,3-b:3’,4’-d]furan and its Radical Cation and Dianion
S. Mori, M. Akita, S. Suzuki, M. S. Asano, M. Murata, T. Akiyama, T. Matsumoto, C. Kitamura, S.-i. Kato.
Chem. Commun. 2020, 56, 5881. DOI: 10.1039/D0CC01638A Highlighted as an inside front cover picture! Highlighted in SYNFACTS
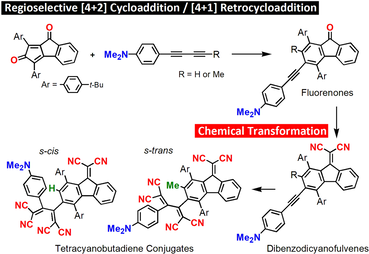
Chemical Transformations of Push–Pull Fluorenones: Push–Pull Dibenzodicyanofulvenes as well as Fluorenone– and Dibenzodicyanofulvene–Tetracyanobutadiene Conjugates
S.-i. Kato, T. Kijima, Y. Shiota, T. Abe, S. Kuwako, H. Miyauchi, N. Yoshikawa, K. Yoshizawa, T. Yoshihara, S. Tobita, Y. Nakamura.
Org. Biomol. Chem. 2020, 18, 4117. DOI: 10.1039/C9OB02706H Highlighted as an outside front cover picture!
Synthesis, Structures, and Properties of Neutral and Radical Cationic S,C,C-Bridged Triphenylamines
S.-i. Kato, T. Matsuoka, S. Suzuki, M. S. Asano, T. Yoshihara, S. Tobita, T. Matsumoto, C. Kitamura.
Org. Lett. 2020, 22, 734. DOI: 10.1021/acs.orglett.9b04575
Reductive Methylation of Triphthaloylbenzene: Isolation and Characterization of Hexamethoxy-trinaphthylene and Two Unexpected Trinaphthylene Derivatives
K. Nishida, K. Ono, S.-i. Kato, C. Kitamura.
Tetrahedron Lett. 2020, 61, 152422. DOI: 10.1016/j.tetlet.2020.152422
Effects of Fluorine Substitution in Quinoidal Oligothiophenes for Use as Organic Semiconductors
K. Yamamoto, S.-i. Kato, H. Zajaczkowska, T. Marszalek, P. W. M. Blom, Y. Ie.
J. Mater. Chem. C 2020, 8, 3580. DOI: 10.1039/C9TC06598A
White-light Emission from a Pyrimidine–Carbazole Conjugate with Tunable Phosphorescence–Fluorescence Dual Emission and Multicolor Emission Switching
T. Ishi-i, H. Tanaka, I. S. Park, T. Yasuda, S.-i. Kato, M. Ito, H. Hiyoshi, T. Matsumoto.
Chem. Commun. 2020, 56, 4051. DOI: 10.1039/D0CC00251H Highlighted as an inside front cover picture!
T. Ishi-i, H. Tanaka, I. S. Park, T. Yasuda, S.-i. Kato, M. Ito, H. Hiyoshi, T. Matsumoto.
Chem. Commun. 2020, 56, 4051. DOI: 10.1039/D0CC00251H Highlighted as an inside front cover picture!
Visualization of Lipid Droplets in Living Cells and Fatty Livers of Mice Based on the Fluorescence of π-Extended Coumarin Using Fluorescence Lifetime Imaging Microscopy
T. Yoshihara, R. Maruyama, S. Shiozaki, K. Yamamoto, S.-i. Kato, Y. Nakamura, S. Tobita.
Anal. Chem. 2020, 92, 4996. DOI: 10.1021/acs.analchem.9b05184
T. Yoshihara, R. Maruyama, S. Shiozaki, K. Yamamoto, S.-i. Kato, Y. Nakamura, S. Tobita.
Anal. Chem. 2020, 92, 4996. DOI: 10.1021/acs.analchem.9b05184
2019
Mechanochromic Fluorescence Based on a Combination of Acceptor and Bulky Donor Moieties: Tuning Emission Color and Regulating Emission Change Direction
T. Ishi-i, H. Tanaka, R. Youfu, N. Aizawa, T. Yasuda, S.-i. Kato, T. Matsumoto.
New J. Chem. 2019, 43, 4998. DOI: 10.1039/C8NJ06050A Highlighted as an outside front cover picture!
T. Ishi-i, H. Tanaka, R. Youfu, N. Aizawa, T. Yasuda, S.-i. Kato, T. Matsumoto.
New J. Chem. 2019, 43, 4998. DOI: 10.1039/C8NJ06050A Highlighted as an outside front cover picture!
2018
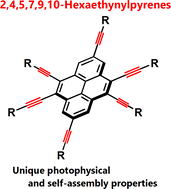
2,4,5,7,9,10-Hexaethynylpyrenes: Synthesis, Properties, and Self-Assembly
S.-i. Kato, H. Kano, K.-i. Irisawa, N. Yoshikawa, R. Yamamoto, C. Kitamura, D. Nara, T. Yamanobe, H. Uehara, Y. Nakamura.
Org. Lett. 2018, 20, 7350. DOI: 10.1021/acs.orglett.8b03290 Highlighted in ChemistryViews
S.-i. Kato, H. Kano, K.-i. Irisawa, N. Yoshikawa, R. Yamamoto, C. Kitamura, D. Nara, T. Yamanobe, H. Uehara, Y. Nakamura.
Org. Lett. 2018, 20, 7350. DOI: 10.1021/acs.orglett.8b03290 Highlighted in ChemistryViews
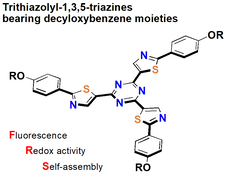
Trithiazolyl-1,3,5-triazines Bearing Decyloxybenzene Moieties: Synthesis, Photophysical, and Electrochemical Properties, as well as Self-Assembly Behavior
S.-i. Kato, S. Jin, T. Kimura, N. Yoshikawa, D. Nara, K. Imakura, Y. Shiota, K. Yoshizawa, R. Katoono, T. Yamanobe, H. Uehara, Y. Nakamura.
Org. Biomol. Chem. 2018, 16, 3584. DOI: 10.1039/C8OB00471D
10-Mesityl-1,8-diphenylanthracene Dimer: Synthesis, Structure, and Properties
A. Shirai, H. Sano, Y. Nakamura, M. Takashika, H. Otani, M. Hasegawa, S.-i. Kato, M. Iyoda.
J. Org. Chem. 2018, 83, 3857. DOI: 10.1021/acs.joc.8b00200
A. Shirai, H. Sano, Y. Nakamura, M. Takashika, H. Otani, M. Hasegawa, S.-i. Kato, M. Iyoda.
J. Org. Chem. 2018, 83, 3857. DOI: 10.1021/acs.joc.8b00200
2017
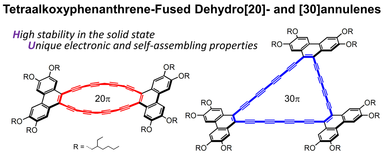
Tetraalkoxyphenanthrene-Fused Hexadecadehydro[20]- and Tetracosadehydro[30]annulenes: Syntheses, Aromaticity/Antiaromaticity, Electronic Properties, and Self-Assembly
N. Takahashi, S.-i. Kato, M. Yamaji, M. Ueno, R. Iwabuchi, Y. Shimizu, M. Nitani, Y. Ie, Y. Aso, T. Yamanobe, H. Uehara, Y. Nakamura.
J. Org. Chem. 2017, 82, 8882. DOI: 10.1021/acs.joc.7b01165 Highlighted in SYNFACTS
N. Takahashi, S.-i. Kato, M. Yamaji, M. Ueno, R. Iwabuchi, Y. Shimizu, M. Nitani, Y. Ie, Y. Aso, T. Yamanobe, H. Uehara, Y. Nakamura.
J. Org. Chem. 2017, 82, 8882. DOI: 10.1021/acs.joc.7b01165 Highlighted in SYNFACTS
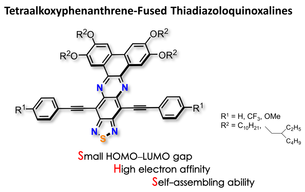
Tetraalkoxyphenanthrene-Fused Thiadiazoloquinoxalines: Synthesis, Electronic, Optical, and Electrochemical Properties, and Self-Assembly
S.-i. Kato, K. Watanabe, M. Tamura, M. Ueno, M. Nitani, Y. Ie, Y. Aso, T. Yamanobe, H. Uehara, Y. Nakamura.
J. Org. Chem. 2017, 82, 3132. DOI: 10.1021/acs.joc.7b00084
Blue Fluorescence from BF2 Complexes of N,O -Benzamide Ligands: Synthesis, Structure, and Photophysical Properties
M. Yamaji, S.-i. Kato, K. Tomonari, M. Mamiya, K. Goto, H. Okamoto, Y. Nakamura, F. Tani.
Inorg. Chem. 2017, 56, 12514. DOI: 10.1021/acs.inorgchem.7b02013
M. Yamaji, S.-i. Kato, K. Tomonari, M. Mamiya, K. Goto, H. Okamoto, Y. Nakamura, F. Tani.
Inorg. Chem. 2017, 56, 12514. DOI: 10.1021/acs.inorgchem.7b02013
2016
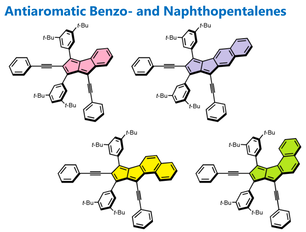
Benzo- and Naphthopentalenes: Syntheses, Structures, and Properties
S.-i. Kato, S. Kuwako, N. Takahashi, T. Kijima, Y. Nakamura.
J. Org. Chem. 2016, 81, 7700. DOI: 10.1021/acs.joc.6b01409
S.-i. Kato, S. Kuwako, N. Takahashi, T. Kijima, Y. Nakamura.
J. Org. Chem. 2016, 81, 7700. DOI: 10.1021/acs.joc.6b01409
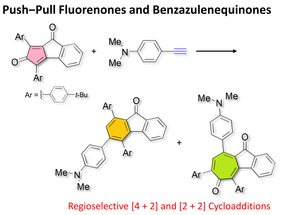
Push‒Pull Fluorenones and Benzazulenequinones: Regioselective [4+2] and [2+2] Cycloadditions of Benzopentalenequinone Derivative and Alkynes Bearing an Aniline Moiety
S.-i. Kato, T. Kijima, Y. Shiota, T. Yoshihara, S. Tobita, K. Yoshizawa, Y. Nakamura.
Tetrahedron Lett. 2016, 57, 4604. DOI: 10.1016/j.tetlet.2016.09.002
Synthesis, Self-Association, and Anion Recognition of Conjugated Macrocycles Composed of Carbazole and Triazolium Moieties
S. Jin, S.-i. Kato, Y. Nakamura.
Chem. Lett. 2016, 45, 869. DOI: 10.1246/cl.160400
Synthesis and Electronic, Optical, and Electrochemical Properties of a Series of Tetracyanobutadiene-Substituted Carbazoles
S.-i. Kato, H. Noguchi, S. Jin, Y. Nakamura.
Asian J. Org. Chem. 2016, 5, 246. DOI: 10.1002/ajoc.201500431
Photochemical Synthesis and Photophysical Features of Ethynylphenanthrenes Studied by Emission and Transient Absorption Measurements
Y. Hakoda, M. Aoyagi, K. Irisawa, S.-i. Kato, Y. Nakamura, M. Yamaji.
Photochem. Photobiol. Sci. 2016, 15, 1586. DOI: 10.1039/C6PP00291A
Y. Hakoda, M. Aoyagi, K. Irisawa, S.-i. Kato, Y. Nakamura, M. Yamaji.
Photochem. Photobiol. Sci. 2016, 15, 1586. DOI: 10.1039/C6PP00291A
2015
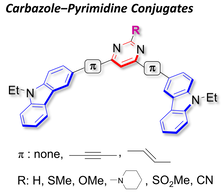
Series of Carbazole‒Pyrimidine Conjugates: Synthesis and Electronic, Photophysical, and Electrochemical Properties
S.-i. Kato, Y. Yamada, H. Hiyoshi, K. Umezu, Y. Nakamura.
J. Org. Chem. 2015, 80, 9076. DOI: 10.1021/acs.joc.5b01409
S.-i. Kato, Y. Yamada, H. Hiyoshi, K. Umezu, Y. Nakamura.
J. Org. Chem. 2015, 80, 9076. DOI: 10.1021/acs.joc.5b01409
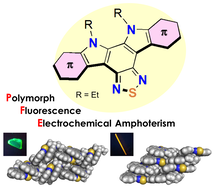
A Series of π-Extended Thiadiazoles Fused with Electron-Donating Heteroaromatic Moieties: Syntheses, Properties, and Polymorphic Crystals
S.-i. Kato, T. Furuya, M. Nitani, N. Hasebe, Y. Ie, Y. Aso, T. Yoshihara, S. Tobita, Y. Nakamura.
Chem. Eur. J. 2015, 21, 3115. DOI: 10.1002/chem.201405478
2014
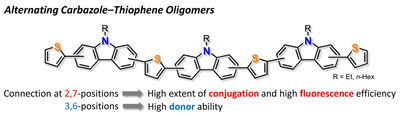
Systematic Structure‒Property Investigations on a Series of Alternating Carbazole‒Thiophene Oligomers
S.-i. Kato, S. Shimizu, A. Kobayashi, T. Yoshihara, S. Tobita, Y. Nakamura.
J. Org. Chem. 2014, 79, 618. DOI: 10.1021/jo402416f
S.-i. Kato, S. Shimizu, A. Kobayashi, T. Yoshihara, S. Tobita, Y. Nakamura.
J. Org. Chem. 2014, 79, 618. DOI: 10.1021/jo402416f
From Homoconjugated Push‒Pull Chromophores to Donor‒Acceptor-Substituted Spiro Systems by Thermal Rearrangement
C. Dengiz, O. Dumele, S.-i. Kato, M. Zalibera, P. Cias, W. B. Schweizer, C. Boudon, J.-P. Gisselbrecht, G. Gescheidt, F. Diederich.
Chem. Eur. J. 2014, 20, 1279. DOI: 10.1002/chem.201303533
2013
Hexadecadehydrodibenzo[20]-, Tetracosadehydrotribenzo[30]-, and Dotriacontadehydrotetrabenzo[40]annulenes: Syntheses, Characterizations, Electronic Properties, and Self-Associations
S.-i. Kato, N. Takahashi, Y. Nakamura.
J. Org. Chem. 2013, 78, 7658. DOI: 10.1021/jo401200m
S.-i. Kato, N. Takahashi, Y. Nakamura.
J. Org. Chem. 2013, 78, 7658. DOI: 10.1021/jo401200m
Tetraalkoxyphenanthrene-Fused Dehydroannulenes: Synthesis, Self-Assembly, and Electronic, Optical, and Electrochemical Properties
S.-i. Kato, N. Takahashi, H. Tanaka, A. Kobayashi, T. Yoshihara, S. Tobita, T. Yamanobe, H. Uehara, Y. Nakamura.
Chem. Eur. J. 2013, 19, 12138. DOI: 10.1002/chem.201301262
Synthesis and Properties of Disiloxane-bridged Cyclophanes Bearing Heteroaromatics
S.-i. Kato, N. Yamazaki, T. Tajima, Y. Nakamura.
Chem. Lett. 2013, 42, 401. DOI: org/10.1246/cl.121271
Synthesis and Properties of Disiloxane-bridged Cyclophanes Bearing Heteroaromatics
S.-i. Kato, N. Yamazaki, T. Tajima, Y. Nakamura.
Chem. Lett. 2013, 42, 401. DOI: org/10.1246/cl.121271
Photochemical Behaviors of a Tethered 1,3-Diketone Derivative Studied by Transient Absorption and Time-resolved EPR Measurements
A. Kobayashi, M. Yamaji, S. Nakajima, K. Akiyama, S. Tero-Kubota, S.-i. Kato, Y. Nakamura.
Chem. Phys. Lett. 2013, 555, 101. DOI: 10.1016/j.cplett.2012.10.067
2012
Bicarbazoles: Systematic Structure-Property Investigations on a Series of Conjugated Carbazole Dimers
S.-i. Kato, H. Noguchi, A. Kobayashi, T. Yoshihara, S. Tobita, Y. Nakamura.
J. Org. Chem. 2012, 77, 9120. DOI: 10.1021/jo3016538
S.-i. Kato, H. Noguchi, A. Kobayashi, T. Yoshihara, S. Tobita, Y. Nakamura.
J. Org. Chem. 2012, 77, 9120. DOI: 10.1021/jo3016538
π-Extended Thiadiazoles Fused with Thienopyrrole or Indole Moieties: Synthesis, Structures, and Properties
S.-i. Kato, T. Furuya, A. Kobayashi, M. Nitani, Y. Ie, Y. Aso, T. Yoshihara, S. Tobita, Y. Nakamura.
J. Org. Chem. 2012, 77, 7595. DOI: 10.1021/jo301458m Highlighted in SYNFACTS
Synthesis and Electronic, Photophysical, and Electrochemical Properties of a Series of Thienylcarbazoles
S.-i. Kato, S. Shimizu, H. Taguchi, A. Kobayashi, S. Tobita, Y. Nakamura.
J. Org. Chem. 2012, 77, 3222. DOI: 10.1021/jo202625p
~2011
Synthesis of [60]Fullerene-Containing [2]Rotaxanes Using Axle Molecules Bearing Donor Moiety
Y. Kasai, C. Sakamoto, N. Muroya, S.-i. Kato, Y. Nakamura.
Tetrahedron Lett. 2011, 52, 623. DOI: 10.1016/j.tetlet.2010.11.155
Y. Kasai, C. Sakamoto, N. Muroya, S.-i. Kato, Y. Nakamura.
Tetrahedron Lett. 2011, 52, 623. DOI: 10.1016/j.tetlet.2010.11.155
Homoconjugated Push‒Pull and Spiro Systems: Intramolecular Charge-Transfer Interactions and Third-Order Nonlinearlities
S.-i. Kato, M. T. R. Beels, P. La Porta, W. B. Schweizer, C. Boudon, J.-P. Gisselbrecht, I. Biaggio, F. Diederich.
Angew. Chem. Int. Ed. 2010, 49, 6207. DOI: 10.1002/anie.201002236
S.-i. Kato, M. T. R. Beels, P. La Porta, W. B. Schweizer, C. Boudon, J.-P. Gisselbrecht, I. Biaggio, F. Diederich.
Angew. Chem. Int. Ed. 2010, 49, 6207. DOI: 10.1002/anie.201002236
Molecular Recognition Properties of Polymethoxybenzenes by Host Molecules Comprised of Two Pyromellitic Diimides and Two Dialkoxynaphthalenes
T. Nakagaki, S.-i. Kato, A. Harano, T. Shinmyozu.
Tetrahedron 2010, 66, 976. DOI: 10.1016/j.tet.2009.11.087
T. Nakagaki, S.-i. Kato, A. Harano, T. Shinmyozu.
Tetrahedron 2010, 66, 976. DOI: 10.1016/j.tet.2009.11.087
Origin of Intense Intramolecular Charge-Transfer Interactions in Non-Planar Push‒Pull Chromophores
S.-i. Kato, M. Kivala, W. B. Schweizer, C. Boudon, J.-P. Gisselbrecht, F. Diederich.
Chem. Eur. J. 2009, 15, 8687. DOI: 10.1002/chem.200901630
S.-i. Kato, M. Kivala, W. B. Schweizer, C. Boudon, J.-P. Gisselbrecht, F. Diederich.
Chem. Eur. J. 2009, 15, 8687. DOI: 10.1002/chem.200901630
Novel Pyromellitic Diimide-Based Macrocycle with a Linear π-Electronic System and Bis(phenylethynyl)pyromellitic Diimide: Syntheses, Structures, Photophysical Properties, and Redox Characteristics
S.-i. Kato, Y. Nonaka, T. Shimasaki, K. Goto, T. Shinmyozu.
J. Org. Chem. 2008, 73, 4063. DOI: 10.1021/jo800283r
S.-i. Kato, Y. Nonaka, T. Shimasaki, K. Goto, T. Shinmyozu.
J. Org. Chem. 2008, 73, 4063. DOI: 10.1021/jo800283r
One-pot Synthesis of Macrocyclic Compounds Possessing Two Cyclobutane Rings by Sequential Inter- and Intramolecular [2 + 2] Photocycloaddition Reactions
H. Miyauchi, C. Ikematsu, T. Shimasaki, S.-i. Kato, T. Shinmyozu, T. Shimo, K. Somekawa.
Tetrahedron 2008, 64, 4108. DOI: 10.1016/j.tet.2008.02.005
H. Miyauchi, C. Ikematsu, T. Shimasaki, S.-i. Kato, T. Shinmyozu, T. Shimo, K. Somekawa.
Tetrahedron 2008, 64, 4108. DOI: 10.1016/j.tet.2008.02.005
The First X-Ray Crystallographic Evidence of a Cyclic Aniline Trimer via Self-Complementary N–H··· π Interactions: The Aniline Inclusion both inside and outside the Macrocyclic Cavity
S.-i. Kato, T. Nakagaki, T. Shimasaki, T. Shinmyozu.
CrystEngComm 2008, 10, 483. DOI: 10.1039/B718306B
S.-i. Kato, T. Nakagaki, T. Shimasaki, T. Shinmyozu.
CrystEngComm 2008, 10, 483. DOI: 10.1039/B718306B
Synthesis, Structural, Spectral, and Photoswitchable Properties of cis- and trans- 2,2,13,13-Tetramethyl- 1,1'-indanylindanes
T. Shimasaki, S.-i. Kato, T. Shinmyozu.
J. Org. Chem. 2007, 72, 6251. DOI: 10.1021/jo0701233
T. Shimasaki, S.-i. Kato, T. Shinmyozu.
J. Org. Chem. 2007, 72, 6251. DOI: 10.1021/jo0701233
Singlet Oxygen Generation by Two-Photon Excitation of Porphyrin Derivatives Having Two-Photon- Absorbing Benzothiadiazole Chromophores
T. Ishi-i, Y. Taguri, S.-i. Kato, M. Shigeiwa, H. Gorohmaru, S. Maeda, S. Mataka.
J. Mater. Chem. 2007, 17, 3341. DOI: 10.1039/B704499B
T. Ishi-i, Y. Taguri, S.-i. Kato, M. Shigeiwa, H. Gorohmaru, S. Maeda, S. Mataka.
J. Mater. Chem. 2007, 17, 3341. DOI: 10.1039/B704499B
Synthesis and Structural and Photoswitchable Properties of Novel Chiral Host Molecules: Axis Chiral 2,2'-Dihydroxy-1,1'-binaphtyl Appended stiff-Stilbene
T. Shimasaki, S.-i. Kato, K. Ideta, K. Goto, T. Shinmyozu.
J. Org. Chem. 2007, 72, 1073. DOI: 10.1021/jo061127v
T. Shimasaki, S.-i. Kato, K. Ideta, K. Goto, T. Shinmyozu.
J. Org. Chem. 2007, 72, 1073. DOI: 10.1021/jo061127v
Supramolecular Assemblies and Redox Modulation of Pyromellitic Diimide-Based Cyclophane via Noncovalent Interactions with Naphthol
S.-i. Kato, T. Matsumoto, K. Ideta, T. Shimasaki, K. Goto, T. Shinmyozu.
J. Org. Chem. 2006, 71, 4723. DOI: 10.1021/jo0600196
S.-i. Kato, T. Matsumoto, K. Ideta, T. Shimasaki, K. Goto, T. Shinmyozu.
J. Org. Chem. 2006, 71, 4723. DOI: 10.1021/jo0600196
Novel 2,1,3-Benzothiadiazole-Based Red-Fluorescent Dyes with Enhanced Two-Photon Absorption Cross-Sections
S.-i. Kato, T. Matsumoto, M. Shigeiwa, H. Gorohmaru, S. Maeda, T. Ishi-i, S. Mataka.
Chem. Eur. J. 2006, 12, 2303. DOI: 10.1002/chem.200500921
S.-i. Kato, T. Matsumoto, M. Shigeiwa, H. Gorohmaru, S. Maeda, T. Ishi-i, S. Mataka.
Chem. Eur. J. 2006, 12, 2303. DOI: 10.1002/chem.200500921
Strongly Red-Fluorescent Novel Donor-π-Bridge-Acceptor-π-Bridge-Donor Type 2,1,3-Benzothiadiazoles with Enhanced Two-Photon Absorption Cross-Sections
S.-i. Kato, T. Matsumoto, T. Ishi-i, M. Shigeiwa, H. Gorohmaru, S. Maeda, Y. Yamashita, S. Mataka.
Chem. Commun. 2004, 2342. DOI: 10.1039/B410016F
S.-i. Kato, T. Matsumoto, T. Ishi-i, M. Shigeiwa, H. Gorohmaru, S. Maeda, Y. Yamashita, S. Mataka.
Chem. Commun. 2004, 2342. DOI: 10.1039/B410016F
Review
Phenanthrylene–Alkynylene Macrocycles, Phenanthrene-Fused Dicyclopenta[b,g]naphthalene, as well as Relevant Diradicaloids and Antiaromatic Compounds
S.-i. Kato.
Adv. Phys. Org. Chem. 2021, 55, 41. DOI: 10.1016/bs.apoc.2021.09.001
S.-i. Kato.
Adv. Phys. Org. Chem. 2021, 55, 41. DOI: 10.1016/bs.apoc.2021.09.001
カルバゾール等の複素環を基盤とするπ共役系化合物の合成と物性
中村洋介,加藤真一郎,野口裕冬.
有機合成化学協会誌, 2013, 71, 779. DOI: 10.5059/yukigoseikyokaishi.71.779
中村洋介,加藤真一郎,野口裕冬.
有機合成化学協会誌, 2013, 71, 779. DOI: 10.5059/yukigoseikyokaishi.71.779
Exohedral Functionalization of Fullerenes and Supramolecular Chemistry
Y. Nakamura, S.-i. Kato.
Chem. Rec. 2011, 11, 77. DOI: 10.1002/tcr.201000036
Y. Nakamura, S.-i. Kato.
Chem. Rec. 2011, 11, 77. DOI: 10.1002/tcr.201000036
Non-Planar Push–Pull Chromophores
S.-i. Kato, F. Diederich.
Chem. Commun. 2010, 46, 1994. DOI: 10.1039/B926601A
S.-i. Kato, F. Diederich.
Chem. Commun. 2010, 46, 1994. DOI: 10.1039/B926601A